Convenience of an oral, targeted treatment
All Revuforj doses should be taken:

orally, twice daily
at about the same time each day
(~12 hours apart)

fasted* or
with a low-fat meal
(~400 calories and
25% or less fat)
In the clinical trial protocol, fasted was defined as at least 2 hours after a meal and 1 hour before the next meal.
Continue Revuforj until disease progression or unacceptable toxicity
MONTHS
For patients without disease progression or unacceptable toxicity, treat for a minimum of 6 months to allow time for a clinical response
Patients are able to take proton pump inhibitors, H2 receptor antagonists, and antacids with Revuforj
Review the drug interactions
Revuforj is available in 3 different tablet strengths, allowing for individualized dosing to meet the needs of your patients
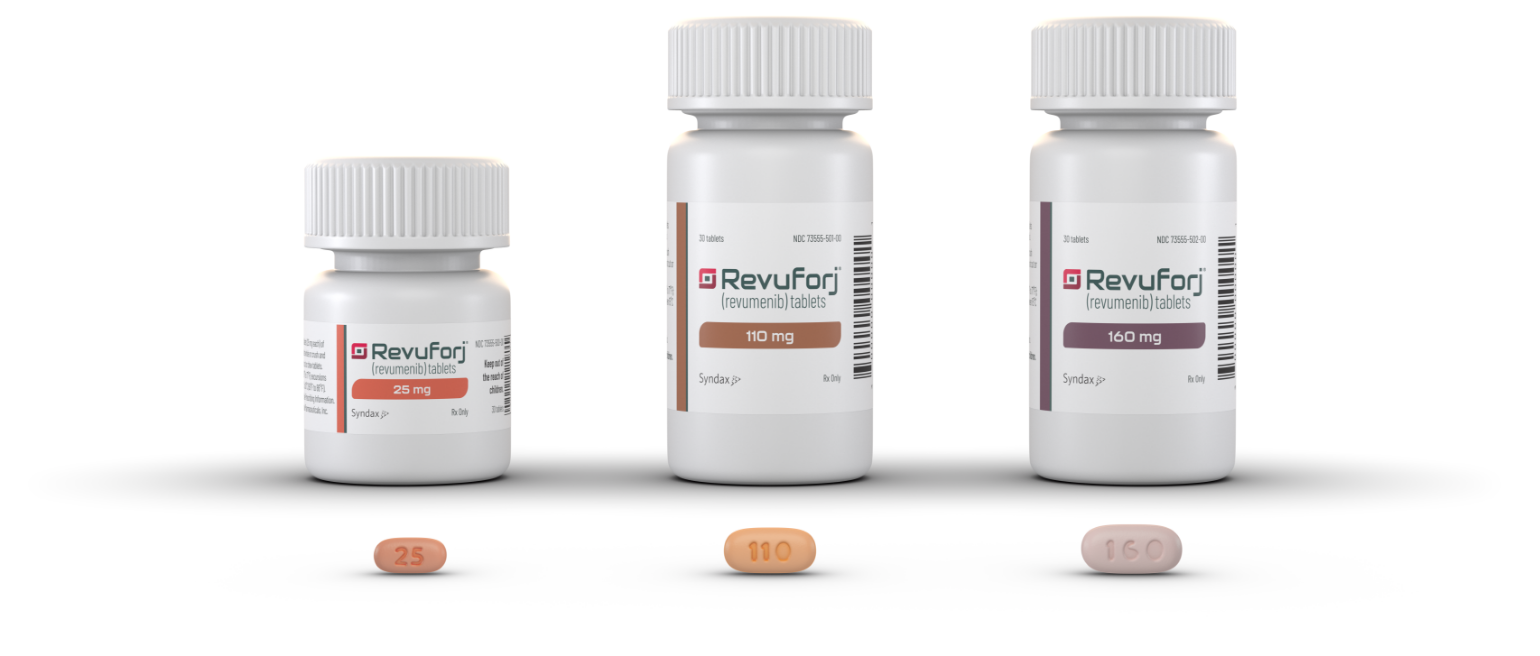
Bottles and tablets shown are not actual size.
-
If needed, attain the desired dose by combining different strengths of Revuforj tablets
-
Advise patients to swallow tablets whole and
to not cut or chew tablets -
If patients are unable to swallow tablets, they may be crushed and dispersed in water and taken within 2 hours of preparation
Review the Instructions for Use with patients and/or their respective caregivers.
Instructions for Use video: How to prepare Revuforj for people who are unable to swallow tablets whole

This video is also available to patients and their caregivers at Revuforj.com
If a dose of Revuforj is missed or not taken at the usual time, administer the dose as soon as possible on the same day and at least 12 hours prior to the next scheduled dose. Return to the normal schedule the following day.
Do not administer 2 doses within 12 hours
Individualized dosing taken orally twice daily
Revuforj dosing recommendations provide guidance for use with and without strong CYP3A4 inhibitors in both adult and pediatric patients
Starting dose and dose modifications for patients 1 year and older
Dose
level
Patients
≥40 kg
Patients
<40 kg*
Starting dose
270 mg BID
160 mg BID
160 mg/m2 BID
95 mg/m2 BID
Reduced dose
160 mg BID
110 mg BID
95 mg/m2 BID
65 mg/m2 BID
BID=twice a day; BSA=body surface area; CYP3A4=cytochrome P450 3A4.
-
See below for the recommended dose and the reduced dose for patients weighing <40 kg with a BSA ≤1.4 m2.
-
If the strong CYP3A4 inhibitor is discontinued, increase the Revuforj dose (after at least 5 half-lives of the strong CYP3A4 inhibitor) to the recommended dosage without strong CYP3A4 inhibitors.
Revuforj dosage for recommended dose and reduced dose
BSA
(m2)
160
mg/m2
95
mg/m2
65
mg/m2
1.4
220 mg
BID
135 mg
BID
100 mg
BID
1.3
220 mg
BID
135 mg
BID
75 mg
BID
1.2
185 mg
BID
110 mg
BID
75 mg
BID
1.1
185 mg
BID
110 mg
BID
75 mg
BID
1
160 mg
BID
100 mg
BID
50 mg
BID
0.9
135 mg
BID
75 mg
BID
50 mg
BID
0.8
135 mg
BID
75 mg
BID
50 mg
BID
0.7
110 mg
BID
50 mg
BID
50 mg
BID
0.6
100 mg
BID
50 mg
BID
25 mg
BID
0.5
75 mg
BID
50 mg
BID
25 mg
BID
0.4
50 mg
BID
25 mg
BID
25 mg
BID
For patients who are unable to swallow Revuforj tablets whole:
Review the Instructions for Use with patients and their caregivers for how to prepare and break apart the Revuforj tablets in water
Monitoring guidance for before and during treatment
Prior to starting Revuforj, reduce the white blood cell count to less than 25 Gi/L
Assess blood counts, electrolytes, and liver enzymes
Perform electrocardiogram (ECG)
prior to the initiation of Revuforj, at least once a week for the first 4 weeks, and at least monthly thereafter
ECG monitoring recommendations for QTc interval prolongation are consistent across the FDA-approved menin inhibitor class
In patients with congenital long QTc syndrome, congestive heart failure, electrolyte abnormalities, or those who are taking medications known to prolong the QTc interval, more frequent ECG monitoring may be necessary.
-
Do not initiate Revuforj in patients with QTcF >450 msec
-
Correct electrolyte abnormalities, including hypokalemia and hypomagnesemia, prior to and throughout treatment with Revuforj
Advise patients to consult their healthcare provider immediately if they feel faint, lose consciousness, or have signs or symptoms suggestive of arrhythmia. Advise patients with a history of hypokalemia or hypomagnesemia of the importance of monitoring their electrolytes.
Verify pregnancy status in females of reproductive potential within 7 days prior to initiating Revuforj
Other drugs may affect the safety and efficacy of Revuforj
Advise patients to inform their healthcare providers of all concomitant medications, including over-the-counter products and supplements.
Concomitant use of
Revuforj with:
Concomitant use of
Revuforj with:
Concomitant use of
Revuforj with:
Effect
Effect
Effect
Action
Action
Action
Strong CYP3A4 inhibitors
Strong or moderate
CYP3A4 inducers
QTc-prolonging drugs
Increases revumenib systemic exposure, which may increase the risk of adverse reactions
May decrease revumenib and increase M1 systemic exposure, which may reduce Revuforj efficacy or increase the risk of QT prolongation associated with the M1 metabolite
May result in an increase in the QTc interval and adverse reactions associated with QTc interval prolongation
If concomitant use of strong CYP3A4 inhibitors is required, reduce the Revuforj dose*
Avoid concomitant use
with Revuforj
Avoid concomitant use with Revuforj†
Revuforj does not have known drug interactions with proton pump inhibitors, H2 receptor antagonists, or antacids
See Section 2.2 of the Full Prescribing Information.
If concomitant use is unavoidable, obtain ECGs when initiating, during concomitant use, and as clinically indicated. Withhold Revuforj if the QTc interval is >480 msec. Restart Revuforj after the QTc interval returns to ≤480 msec.
Doses can be modified as needed to help manage adverse reactions
Select an adverse reaction below to see the recommended action
Recommended action:
-
If differentiation syndrome is suspected, administer systemic corticosteroids and initiate hemodynamic monitoring until symptom resolution and for a minimum of 3 days
-
Interrupt Revuforj if severe signs and/or symptoms persist for more than 48 hours after initiation of systemic corticosteroids, or earlier for life-threatening symptoms such as pulmonary symptoms requiring ventilator
support. Resume Revuforj at the same dose when signs and symptoms improve to Grade 1* or lower
Recommended action:
-
Initiate treatment with hydroxyurea in patients with an elevated or rapidly rising leukocyte count. Add leukapheresis if clinically indicated
-
Taper hydroxyurea only after leukocytosis improves or resolves
Recommended action:
-
Interrupt Revuforj
-
Check electrolyte levels. Correct hypokalemia and hypomagnesemia
-
Restart Revuforj at the same dose level after the QTc interval returns to less than or equal to 480 msec
Recommended action:
-
Interrupt Revuforj
-
Check electrolyte levels. Correct hypokalemia and hypomagnesemia
-
Restart Revuforj at the reduced dose level† after the QTc interval returns to less than or equal to 480 msec
Recommended action:
-
Permanently discontinue Revuforj
Recommended action:
-
Supplement potassium and/or magnesium
-
Continue Revuforj
Recommended action:
-
Supplement potassium and/or magnesium, and recheck levels within 24 hours
-
On recheck of potassium and magnesium labs within 24 hours, if potassium is greater than 3.5 mEq/
L and/or magnesium is greater than 1.6 mg/dL, continue Revuforj. If potassium is less than 3.5 mEq/L and/or magnesium is less than 1.6 mg/dL, hold Revuforj and continue supplementation; resume Revuforj at the same dose level when
the correction is complete
Recommended action:
-
Interrupt Revuforj until recovery to Grade 1* or baseline
-
If recovered in ≤7 days, restart Revuforj at the same dose level. If the same Grade ≥3* toxicity recurs, interrupt Revuforj until recovery to Grade 1* or baseline. Restart Revuforj at the reduced dose level†
-
If recovered in >7 days, restart Revuforj at the reduced dose level.† If the same Grade ≥3* toxicity recurs, discontinue Revuforj
Recommended action:
-
Interrupt Revuforj until recovery to Grade ≤2* or baseline
-
Restart Revuforj at the same dose level
-
If Grade 4* neutropenia or thrombocytopenia recurs without attributable cause, interrupt Revuforj until recovery
to Grade ≤3.* Restart Revuforj at the reduced dose level†
Recommended action:
-
Permanently discontinue Revuforj
Other actions may be necessary based on your clinical judgment.
Grade 1 is mild, Grade 2 is moderate, Grade 3 is severe, Grade 4 is life-threatening. Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE version 5.0).
See Tables 4, 5, and 6 in the Full Prescribing Information for the reduced dose levels.


